

OrpheEdge + OrpheLink: Building a Secure, Centrally Managed Data Backbone
Background & Challenges
n the digitalization of cross-border healthcare, it is essential to securely and efficiently transfer highly sensitive patient data, including 3D scan files, X-ray images, and orthodontic designs. For dental manufacturers operating between Taiwan and Southeast Asia (such as Vietnam, Thailand, and the Philippines), the challenge is especially pressing. Traditional methods that rely on public cloud or third-party platforms not only expose data to external risks but also struggle to meet the privacy and compliance requirements of different countries.
Solution: OrpheEdge + OrpheLink
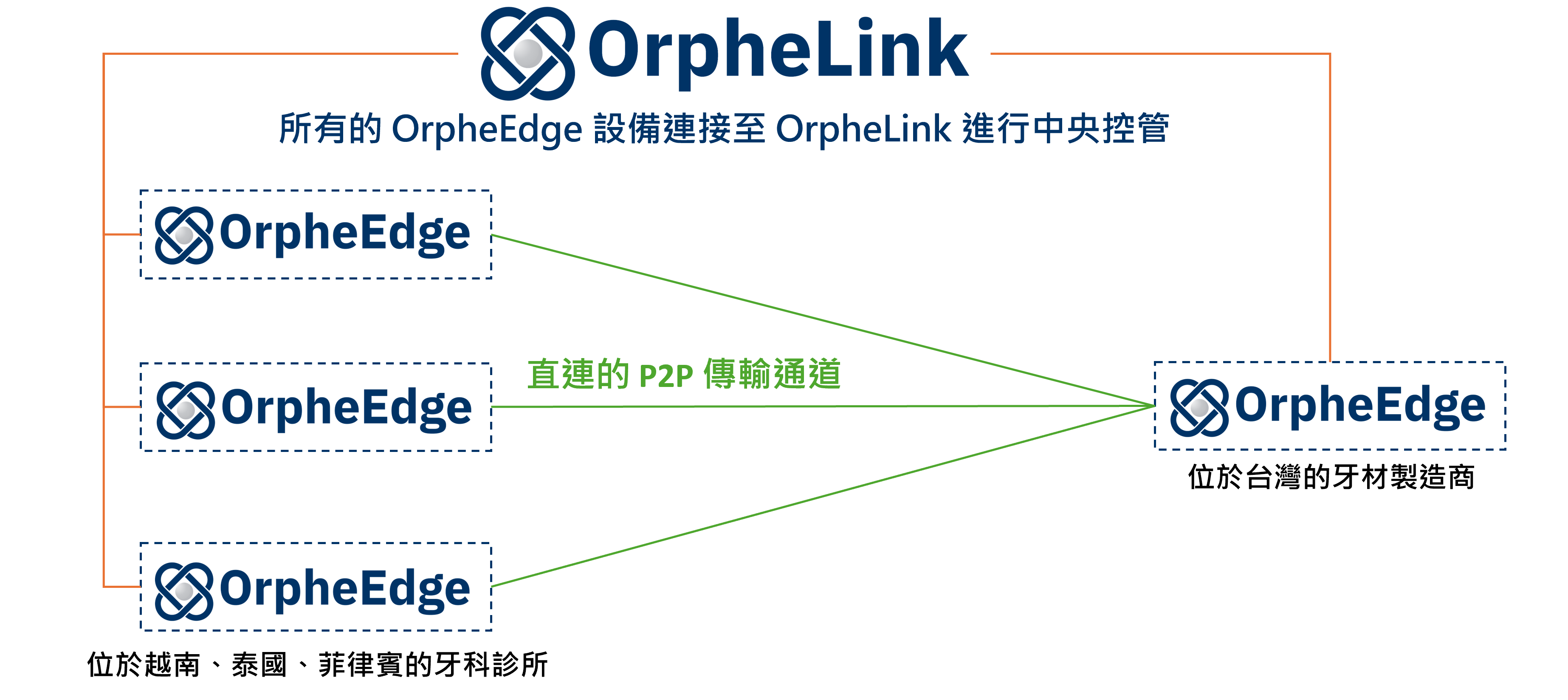
To address these issues, the dental manufacturer deployed OrpheEdge hardware nodes between its clinics and laboratories in Taiwan. This established secure, plug-and-play P2P tunnels, eliminating the need for complex VPN or cloud configurations. All data is transmitted directly with end-to-end encryption.
At the same time, through OrpheLink, the IT team can easily manage multiple clinic nodes from a single interface, monitor status, and configure permissions—making cross-border operations far simpler and more secure.
| Category | Before (Traditional) |
After (OrpheEdge + OrpheLink) |
|---|---|---|
| Connection | Public cloud or third-party relay platforms | OrpheEdge establishes secure P2P tunnels — no cloud or relays required |
| Security | Sensitive data exposed to external networks | Fully end-to-end encrypted, deployed invisibly |
| Deployment | Repeated IT setup and maintenance required | OrpheEdge plug-and-play; clinics go live within minutes |
| Management | Difficult to centrally manage multiple sites | OrpheLink provides unified configuration and monitoring |
| Compliance | Cross-border data flow difficult to regulate | Encrypted transfers meet compliance with no intermediary exposure |
Results & Extended Value
This architecture enables local dental manufacturers in Taiwan and clinics across Southeast Asia to securely and efficiently exchange clinical data, protecting patient privacy while complying with both Southeast Asian and Taiwanese healthcare data regulations. Beyond the dental sector, this model can be quickly replicated in other industries that require cross-border data exchange—such as medical imaging, clinical trials, and even R&D collaboration in manufacturing.